121
Solution:
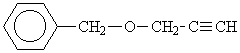
Reason:
With the solution of this question another possibility of the way A is to be used ( see step 120):The compound contains a phenyl group of C6H5. Thus C4H5O must be looked up for univalent remainder, fitting the spectrum. The small number of H-atoms suggests that multiple bonds and thus possibly "long range" couplings.
- Predicates from the chemical shifts of the signals:
d in ppm Possible groupings 2,3 (triplet) -CH3 (aromatically) CH3-CO- CH3-C║C- -CH2-C=C -CH2-CO- -OH H-C║C 4,1(Dublett) CH3O- -CH2O- =CH-O- -OH 4,6 =CH-O- -CH2O- -OH =C=CH2 - Analysis of intensities and couplings:
- The multiplicity of the signals with d = 2.3 ppm and d = 4.1 ppm shows that CH2-
group with a CH-group couples: HC- Ę Ę Ę Ę
-CH2- .
The intensities acknowledge this findings.
From the structures possible for the first signal is applicable thus only C║C-. Altogether we maintain: HC║C-CH2-O-. - The singlet signal with d = 4.6 ppm with same intensity as the Dublett can come then only from not coupling CH 2 - group.
- The multiplicity of the signals with d = 2.3 ppm and d = 4.1 ppm shows that CH2-
group with a CH-group couples: HC- Ę Ę Ę Ę
-CH2- .
- Check on the basis the sum formula:
Since the univalent remainder contains only four C-atoms, the following structure corresponding with the spectrum comes into consideration: HC║C-CH2-O-CH2-
Solve the next question now.