36
Sorry, wrong answer!
Have another, closer look at the isomer and follow the approach out-lined on page 19:One option is to analyze the remainder of the molecule from the view point of each single proton, for example from H1 and H3:
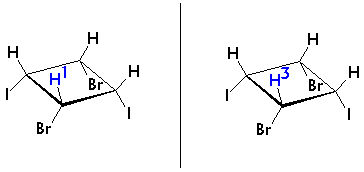
H1 and H3 obviously have entirely identical environments. They should give rise to a single signal.
Alternatively you can make use of the symmetry of the molecule.
Did you notice that this molecule has an axis of two-fold rotational symmetry (C2) ?
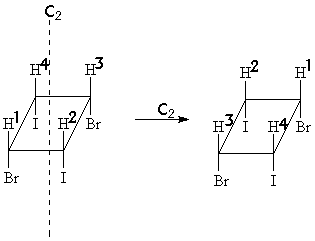
Therefore H1 and H3 exchange positions upon a 180° rotation around (C2)!
| Continue this analysis and decide which protons
are equivalent, as well as how many signals you can expect in the
spectrum of this isomer! When you are done, move on to page 39! |