76
Answer:
The reasoning is analogous to the one for proton A, i.e. the signal of proton X is also a doublet.In the external magnetic field B0 the energy levels of the proton X split into two, and transitions between them become possible, giving rise to resonance signals.
Due to the coupling to the two possible spin alignments of the proton A there are a total of four possible energy levels, and the number of possible transitions doubles. Since the two transitions of a doublet have almost identical energy differences, they occur with the same probabilities, and the ratio of their intensities is 1 : 1.
Another example:
| In bromo-acetaldehyde |
|
In an external magnetic field B0 the energy of the proton A is initially split into two levels.
The spins of the two protons HX of the neighboring CH2Br group can form the following spin combinations in relation to the external field:
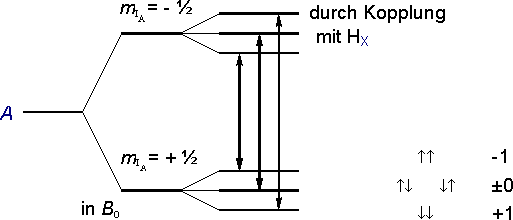 |
!!! |
Due to the coupling between any of the three possible spin combinations for the pair of protons X and the proton A, each of the two energy levels of A is split up into three sublevels. Instead of just one transition between the two energy levels of A, now three closely spaced transitions are possible.
(Triplet: nx = 2, MA = 3).
The ratio of the intensities of the individual triplet lines is 1 : 2 : 1, since out of the four possible spin combinations for the pair X two will have a total spin of 0 (Ż and Ż).
| What is the fine structure of the signal of the
two protons X? Compare your answer to the solution on page 78! |